FG Commerce/E+ through Getty Photographs
Background, key-value driver for BTAI is in BXCL501, and the upcoming PDUFA is the important thing catalyst to give attention to
We’re initiating protection on BioXcel Therapeutics (NASDAQ:BTAI) with a speculative-BUY score with a USD 126 goal worth. Our principal funding thesis revolves across the PDFUA of BXCL501 (sublingual dexmedetomidine [alpha-2 adrenergic receptor agonist]). BXCL501 has a PDFUA date scheduled for April fifth for bipolar and schizophrenia-related agitation. If accepted, we count on peak gross sales round USD 800-1Bn (risk-adjusted). We imagine the present share worth is undervalued and the inventory is buying and selling as if BXCL501 won’t obtain the FDA’s stamp of approval.
BTAI IR Presentation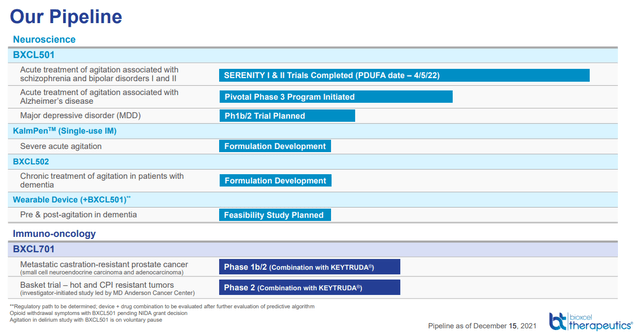
3 Month delay doesn’t concern us, we count on the drug to be accepted with no downside
BTAI IR presentation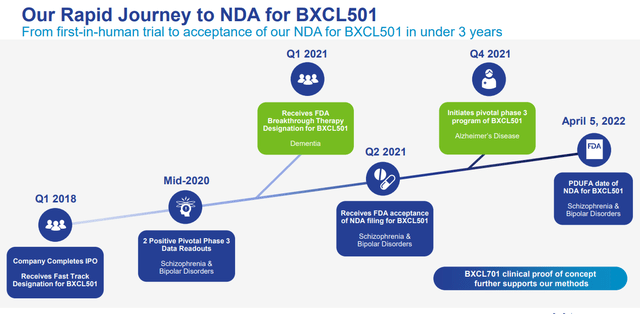
On December 2021, BTAI introduced that the FDA had delayed the NDA for BXCL501 for acute therapy of agitation in schizophrenia and bipolar dysfunction by three months, now, the brand new PDUFA date is April fifth, 2022. After the information, the corporate has introduced that 1) the US FDA didn’t request extra knowledge, 2) the extension is because of FDA requiring extra time for reviewing BTAI’s scientific knowledge.
These are three the explanation why we’re nonetheless optimistic concerning the upcoming PDUFA outcomes, 1) the sturdy SERENITY-1 and a couple of Part III knowledge and the excessive unmet want within the area, 2) the truth that there was no full response letter issued (CRL normally signifies that the FDA is detrimental concerning the drug and as such, significantly lowers the chance of approval), 3) with the brand new FDA commissioner Robert M. Califf M.D named on Feb 2021, we imagine there may be extra regulatory readability throughout the sector.
Illness background: high-unmet want with a clearly outlined affected person inhabitants
BTAI IR presentation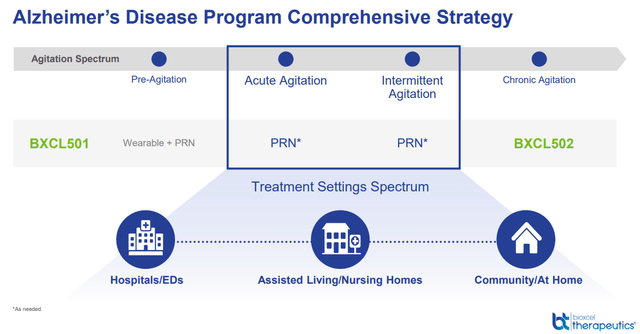
Agitation is a typical difficult-to-treat symptom in most neuropsychiatric situations.
At present, there is no such thing as a universally accepted definition as to what constitutes as agitation, though current makes an attempt have been made to achieve a consensus for agitation in dementia and cognitively impaired sufferers. In schizophrenia and bipolar populations, a broad vary of options have been described together with bodily or psychological unease, interior stress, restlessness, irritability, pleasure, uncooperativeness, anxiousness, a motor exercise that’s extreme, inappropriate or purposeless, and motor stress [1,2,3,4]. Importantly, though aggressive and violent conduct generally happen, professional opinion asserts that agitation is distinct from aggression… Supply
In our view, we imagine the drug shall be extremely helpful within the emergency rooms, greater than within the outpatient setting. We imagine the market alternative is extremely compelling, even contemplating the corporate’s restricted (or unproven) skill to market the product for the primary time.
In keeping with the corporate, over 150M individuals globally with schizophrenia, bipolar dysfunction, dementia, delirium and opioid use dysfunction. Moreover, over 13M sufferers within the U.S. expertise agitation inside these illness areas and greater than 200M agitation episodes per 12 months within the U.S, representing a multi-billion-dollar healthcare burden. Supply
The present SOC panorama signifies a excessive unmet want
Biotechvalley Insights Analysis, Up-to-date tips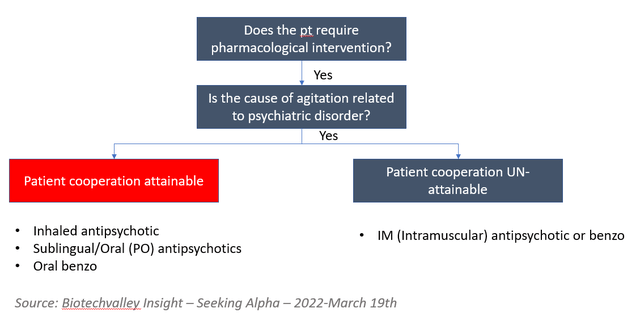
At present, the therapy choices are restricted as the one choices are 1) bodily restraining sufferers, 2) antihistamines, 3) antipsychotics, and benzodiazepines (to over-sedate sufferers). For mildly agitated sufferers, antihistamines (Benadryl or Vistaril) are given, after that, if it does not assist, benzodiazepines or antipsychotics are used. Antihistamines are used 1st line for gentle circumstances as they’re moderately efficient (although not efficient as benzo or antipsychotics), decently fast onset of motion, significantly safer than benzo and antipsychotics, and are usually not addictive (not like Benzos). For acute sufferers, particularly amongst these the place affected person coordination is unattainable, intramuscular benzo or antipsychotics are used as a first-line. Benzodiazepines (i.e., Lorazepam or Diazepam) is extremely addictive and problematic for the aged inhabitants as they will precipitate falls. On the antipsychotic medicine entrance, we spotlight that the category obtained a black-box warning from the FDA for aged populations.
Three years after the Meals and Drug Administration (FDA) instituted a black-box warning for all second-generation antipsychotic (SGA) medicines about elevated danger of loss of life in aged dementia sufferers, an analogous warning is being added to the labels of first-generation antipsychotics (FGAs) similar to haloperidol and perphenazine. – Supply
Moreover, antipsychotics may cause many disagreeable side-effects, similar to acute dystonia (acute muscle cramps), which may even result in cramps within the diaphragm, stopping the sufferers from respiration. One other downside is that antipsychotics do not at all times work effectively on sufferers with out schizophrenia, making it a restricted possibility for prescribers.
To summarize, the important thing unmet want on this area could be:
1) Efficient drug with a quick onset of motion, 2) short-term and long-term security, 3) non-addictive, 4) handy (non IM or IV), which in our view, BXCL501 addresses.
Apart from efficacy, security, the onset of motion benefits, we spotlight the comfort that sublingual or buccal dosage kind can provide sufferers as it may be self-administered by sufferers and the onset of motion is quicker than different choices obtainable. This can be a vital benefit over painful intramuscular SOC therapies (IV therapies are usually not readily used because of their cumbersome nature to deal with agitation) and the drug has the potential to be taken prophylactically to forestall agitations. Particularly in an emergency setting, we spotlight that many sufferers don’t give consent to IM dosing and it is vitally tough to deal with these sufferers and stop them from hurting themselves or hurting others. As such, BXCL501 will present a wonderful approach to defend the affected person’s private autonomy and provide a extra patient-friendly therapy possibility (easy route of administration). We expect the drug can cowl 60-70% of the affected person inhabitants in an ER setting, who’ve mild-moderately agitated/with anxiousness (who do not must be restrained or require extra robust sedation), which is a sizeable inhabitants globally.
BXCL501 has a novel mechanism of motion: Selective alpha-2a receptor agonist
BXCL501 has a novel mechanism of motion clearly differentiated from different SOCs.
BTAI IR deck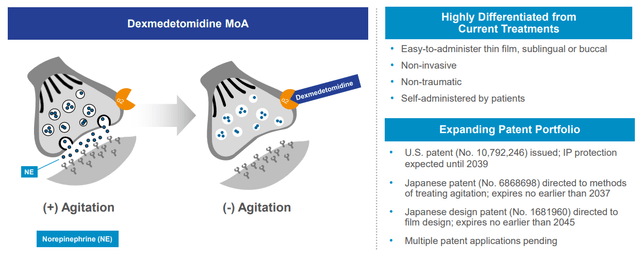
Scientific knowledge is extremely compelling to disregard
We just like the scientific knowledge because the drug’s therapy results in schizophrenia and bipolar sufferers with acute agitations proven to be 1) quick, 2) excessive diploma of efficacy, 3) sturdy, 4) handy (SC supply), 5) secure and tolerable. As well as, we imagine the trial designs (described under) have been sturdy and correctly powered (randomized, double-blinded, placebo-controlled parallel-group adaptive trials in a complete of 759 sufferers).
Schizophrenia: Part III – SERENITY I
The SERENITY I enrolled 381 affected person inhabitants with agitation related to schizophrenia/schizoaffective dysfunction. Arms have been randomized, and sufferers obtained ‘501 120 micrograms, 180 micrograms, or placebo. The trial’s key main endpoint was the discount in acute agitation measured by the Optimistic and Unfavourable Syndrome Scale (change in Excitatory Element (PEC) from baseline in comparison with placebo). The secondary endpoint studied seemed on the earliest time the place an impact on agitation is clear as measured by the change from baseline in PEC complete rating.
Bipolar Dysfunction: Part III – SERENITY II Trial
On the design entrance, the SERENITY II trial enrolled 378 sufferers who’ve agitation related to bipolar problems. Three therapy arms have been randomized to obtain two totally different investigative medicine, ‘501 of 120 mcg and 180 mcg, and the third arm was a placebo arm. The first endpoint of the research was a discount in acute agitation, measured based mostly on Optimistic and Unfavourable Syndrome Scale (Excitatory Element change vs placebo). The secondary endpoint that the research checked out was a change from baseline in PEC complete rating.
Compelling efficacy knowledge
The research confirmed statistically vital and clinically significant reactions with regard to constructive and detrimental syndrome scale, excitatory part rating at two hours and likewise sufferers met secondary endpoints, confirming the information from the first endpoints. In each research, we spotlight that sufferers who obtained each doses (high and low doses) have been in a position to meet main endpoints. We imagine the drug is efficient, speedy, and confirmed sturdy agitation enhancements which are superior to placebo (measured by the constructive and detrimental syndrome scale, PEC, ACES, and Scientific International impression (CGI-I enhancements).
We imagine security knowledge to be the important thing selling-point for the drug.
Additionally, we spotlight the drug confirmed robust security and tolerability with no severe adversarial occasions reported and the most typical adversarial occasions have been quite minor, together with somnolence (21% (120mcg dose) and 22% (180mcg) vs 6% management arms), dizziness, dry mouth, which does not fear us. Even on the somnolence arm, we spotlight that 75% of sufferers had gentle somnolence which is considerably higher than the usual of care antipsychotics and sedatives that causes a excessive diploma of sedation. We observe that contemplating the drug acts on alpha 2 adrenergic receptor, BXCL501 exacerbated extrapyramidal signs and respiratory despair, that are additionally current in first-line therapy for these targets. Moreover, we spotlight the drug had a really low degree of discontinuation because of adversarial occasions including extra confidence to the drug’s security knowledge, SERENITY I, 0 for 180 mcg dose and a couple of sufferers for 120 mcg dose and 0 pt for the placebo arm. Through the SERENITY II trial 0, 1, and 0 pts have been discontinued respectively.
R-X Recordsdata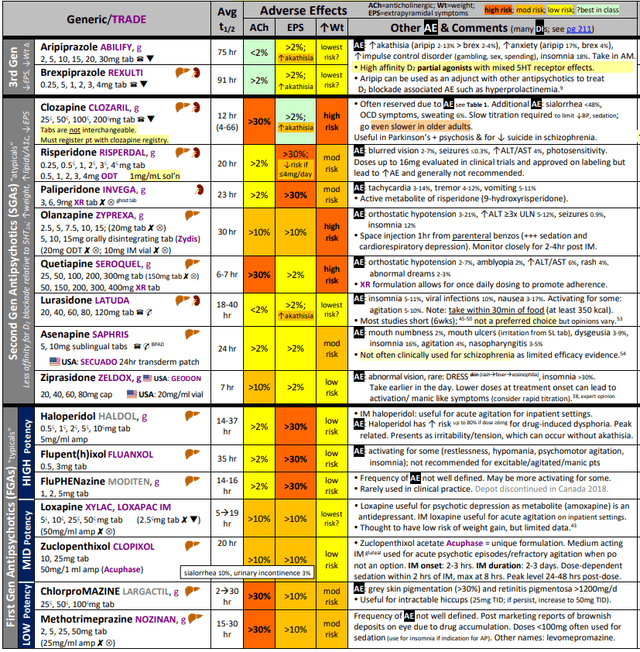
Valuation: TP of USD 123
BMT knowledge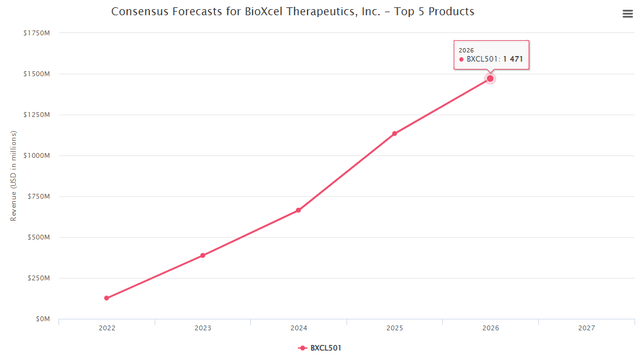
The consensus forecast for BXCL501 is round USD 1.4 Bn (in keeping with BioMed-Tracker’s consensus knowledge). If we risk-adjust the chance of approval for psychiatry illness (91%), we get to a risk-adjusted peak gross sales variety of 1.27Bn. Nevertheless, to train warning because the drug is focusing on a really fragmented geriatric care market, we used USD 800M-1bn peak gross sales for valuation, and a peak gross sales a number of of three is acceptable. As such, we imagine the suitable valuation for the BXCL501 asset must be round USD 3B.
Biomedtracker – POS for BLA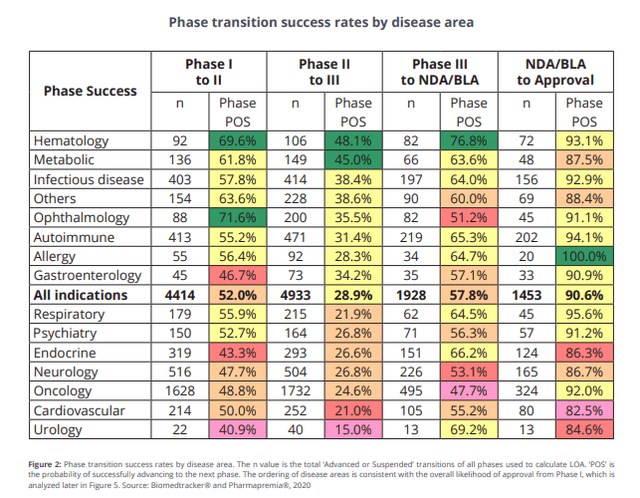
Utilizing the corporate’s present enterprise worth of USD 239M and 232M money (Determine under), we imagine the corporate’s acceptable goal worth must be round USD $123 per share, which represents nearly 600-700% upside from the present share worth.
Searching for Alpha
Hospital formulary entry stays to be the important thing focus for business uptake
We expect the drug wouldn’t have an excessive amount of bother stepping into hospital formularies contemplating the drug’s sturdy scientific knowledge and the truth that different SOC medicine are dangerous. Nevertheless, hospital formulary entry could also be powerful, and with out correct reimbursement, we predict the business launch could also be tough for the corporate, for instance, Alexza Pharmaceutical’s Adasuve (inhaled antipsychotic) has obtained approval in 2012 and was marketed by TEVA Pharmaceutical (TEVA) for agitation in bipolar and schizophrenia, although the drug initially guided peak gross sales to be above USD 200M, however disillusioned buyers with are its business efficiency because of issues of safety (primarily bronchial spasms – that result in REMS) that prevented it from getting acceptable formulary entry. Nevertheless, contemplating the undoubtedly clear security profile of ‘501, we imagine the drug will be capable of obtain favorable formulary entry.
Apart from bipolar and schizophrenia, we spotlight that the corporate can be focusing on agitation associated to dementia (with a considerably bigger market dimension than the 2 aforementioned indications), which in our view, the market is disregarding.
Dangers
1. Uncertainty round its label stays – relying on the institutional or broad setting the market potential shall be considerably modified. Additionally, commercialization is a unique ball-game in comparison with drug growth, and there’s a chance that the corporate under-delivers because of lack of business functionality.
2. Regulatory Danger: FDA might not enable the drug’s approval, and the inventory can fall again to its money worth (USD ~7 per share).
3. Macro-risk, the battle between Ukraine and Russia might plague the inventory market and strain the pre-commercial biotech (XBI)(IBB) market shifting ahead.
4. Growing rates of interest might negatively impression the inventory’s efficiency.
Conclusion
We just like the BXCL501’s section III knowledge, and we imagine the drug has a really excessive probability of getting accepted throughout the PDUFA date. We imagine the present market worth (17.66 USD – as of March nineteenth) to be a particularly engaging entry level for buyers because the inventory obtained over punished because of common macro pressures – rising rates of interest and geopolitical battle between Ukraine and Russia (i.e., speculative tech (ARKK), biotech (XBI), crypto-collectibles (BTC-USD) (ETH-USD) has collapsed). With the battle in Ukraine (probably) reaching its closing levels (or is absolutely priced in into the market) and with FED’s current announcement on its plans on rising the rate of interest, we imagine it’s time to begin shopping for beaten-down high quality biotech names like BioXcel. We spotlight that the corporate has a powerful stability sheet (possible need not increase once more anytime quickly) with USD 232M money, and we like the truth that the corporate has been investing in its business capabilities. We concede that relying on the label, institutional targeted or retail pharmacy targeted launch will impression the height gross sales vastly, however uncertainty across the label however, we imagine the present worth degree is just too engaging to disregard. At this level, we imagine the bull-case (USD 126, PDUFA approval with broader indication) to be (+600%) and the draw back is restricted to the corporate’s money worth (USD 7 per share -60% draw back), making a wonderful risk-reward level for speculative buyers to ascertain a place. Now we have been slowly accumulating a sizeable place and planning to purchase dips as we get nearer to the PDUFA date.


