Modern biotechnology places a significant emphasis on understanding the structure and function of proteins. Understanding three-dimensional (3D) structures at atomic resolution help in molecular docking, structure-based drug designing, and understanding how proteins work.
The function and structure of proteins are widely studied in proteomics, which facilitates the recognition of protein biomarkers associated with specific disease states, thereby providing potential targets for therapeutic treatment.
Knowledge about the 3D structure of proteins helps in gaining a higher understanding of how the protein works and how to use that collective data for various purposes.
At present, cryo-electron microscopy (EM), small angle X-Ray scattering (SAXS), nuclear magnetic resonance (NMR) spectroscopy, and X-Ray crystallography are used extensively in efforts to describe and understand molecular structures and molecular recognition mechanisms.
Here, the expanding applications and benefits of the SAXS technology in 3D protein structure analysis have been discussed.
Why is the SAXS technique used in 3D protein structure analysis?
A biophysical method used to acquire knowledge of the overall shape and structural transitions of biological macromolecules in a solution is called the SAXS technique. It provides a piece of low-resolution information on the shape, conformation, and assembly state of proteins, nucleic acids, and various other macromolecular complexes.
SAXS is a rapid technique and time-resolved study yielding unique information about the kinetics of processes and interactions, especially by using high-intensity synchrotron sources.
Since proteins are found in intricate biological samples, protein complex analysis involves a complete evaluation of the structure and function of proteins. The structure of protein complexes can be easily discovered using contemporary protein complex analysis methods.
SAXS is one of the few methods that allows quantitative characterization of conformational polydispersity of completely or partially disordered macromolecules, including multi-domain proteins with flexible linkers and intrinsically disordered proteins (IDPs).
The growth in the global 3D protein structure analysis market is expected to be driven by technological advancements in equipment for protein structure analysis and by an increase in R&D expenditure to accelerate protein structure analysis.
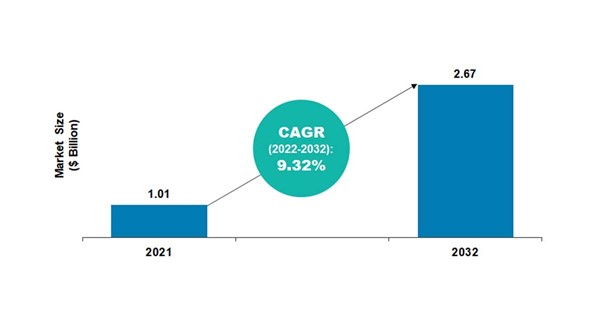
According to BIS Research, the global 3D protein structure analysis market was valued at $1.01 billion in 2021, and it is expected to reach $2.67 billion by 2032, growing at a CAGR of 9.32% during the forecast period 2022-2032.
Find more details on this report in this FREE sample.
Principles, Benefits, and Limitations of SAXS
SAXS technique holds quite a simple and relative principle. A monodispersed prepared solution of macromolecule(s) of interest is placed within a capillary exposing a high-energy monochromatic X-Ray beam.
These photons of the X-Ray beam cause electrons of the atomic shell to begin oscillation which results in a spherical wave being emitted of the same energy and wavelength. Since each electron oscillates, a constant background is achieved, with a resulting electron density of the macromolecule to contrast with the background.
SAXS does not require any special sample preparation and can be performed in almost any buffer, and has the ability to provide information about flexible regions of a macromolecule. SAXS technique is often used to confirm the static structures obtained from the X-Ray imaging spectrometer (XRC) are consistent in solution. SAXS technique requires a relatively small amount of sample investment (typically 50-100 µL) and a relatively small amount of experiment time, i.e., 30 minutes to one hour.
The vulnerability to sample aggregation or degradation leading to incorrect structural predictions is considered to be one of the biggest limitations of the SAXS technique. While sample degradation leads to underestimating molecular properties, an aggregation of even 5% can scatter light in a huge amount, particularly leading to an overestimation of the maximal particle dimension (Dmax) and radius of gyration (Rg).
Also, distortion of the data could occur during the potential for radiation damage to the sample during exposure. A comparison of sample quality before and after SAXS sample exposure can be made to ensure that the end data is accurate.
Conclusion
The global 3D protein structure analysis market is a well-established market and is expected to grow further in the coming years due to the continuous technological advancements in equipment for protein structure analysis, along with the increase in the use of the SAXS technique for 3D protein structure analysis.
Interested to know more about the developing technologies in your industry vertical? Get the latest market studies and insights from BIS Research. Connect with us at [email protected] to learn and understand more.


